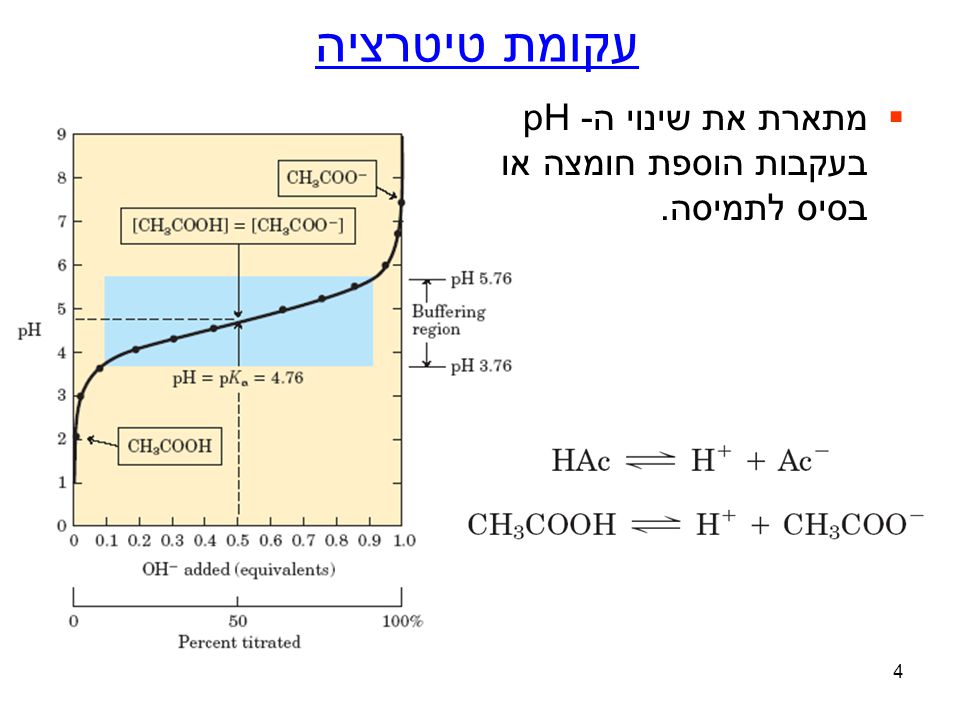
u05de\u05d0\u05d6\u05df_\u05d7\u05d5\u05de\u05e6\u05d9_\u05d1\u05e1\u05d9\u05e1\u05d9_\u05d5\u05ea\u05de\u05d5\u05e0\u05ea_\u05d2\u05d6\u05d9\u05dd_\u05d1\u05d3\u05dd_8860.pptx - \u202b\u05de\u05d0\u05d6\u05df \u05d7\u05d5\u05de\u05e6\u05d9\u202a ...

Optimized Procedure for Determining the Adsorption of Phosphonates onto Granular Ferric Hydroxide using a Miniaturized Phosphorus Determination Method | Protocol (Translated to Hebrew)

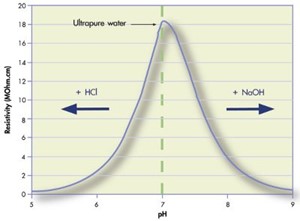
חזרה לקורס מכינה.doc - 3M 100" H2SO4 .1 200" NaOH 1.6M : .PH < 7 2. PH > 7 3. PH = 7 4 .1 .2 : MnCl2(s) + Cl2(g) + 2H2O(l )(4HCl(g) + MnO2(s 4 | Course Hero

חזרה לקורס מכינה.doc - 3M 100" H2SO4 .1 200" NaOH 1.6M : .PH < 7 2. PH > 7 3. PH = 7 4 .1 .2 : MnCl2(s) + Cl2(g) + 2H2O(l )(4HCl(g) + MnO2(s 4 | Course Hero